|
If a gas is given off, note the color and odor of the gas. This is probably water, indicating that the compound is a hydrate. Heating a compound can cause a liquid to condense on the inside of the test tube. The following is a table of the colors of metal ions in solution with NO 3. 
Ionic compounds formed from the representative elements tend to be white or colorless, while ions of transition elements tend to be colored. Note the color and shape of the crystals. The compound will most likely be in solid form. The basic testing procedure for identifying a salt is as follows. There are two general situations in which qualitative analysis is used - in the identification of a simple salt, or the identification of multiple cations in a solution. 
If no precipitate is formed when the reaction is run, then ion A is clearly not present in the unknown solution and a different reaction will have to be run to determine what ions are present. For example, if a reaction is known to produce a precipitate if ion A is present and a precipitate is formed when the reaction is run, then ion A may be present in solution (there may be, and usually are, other ions that will also precipitate with a particular reagent). By predetermining what the particular reaction will produce if a specific ion is present, the ions that actually are in the solution can be identified. 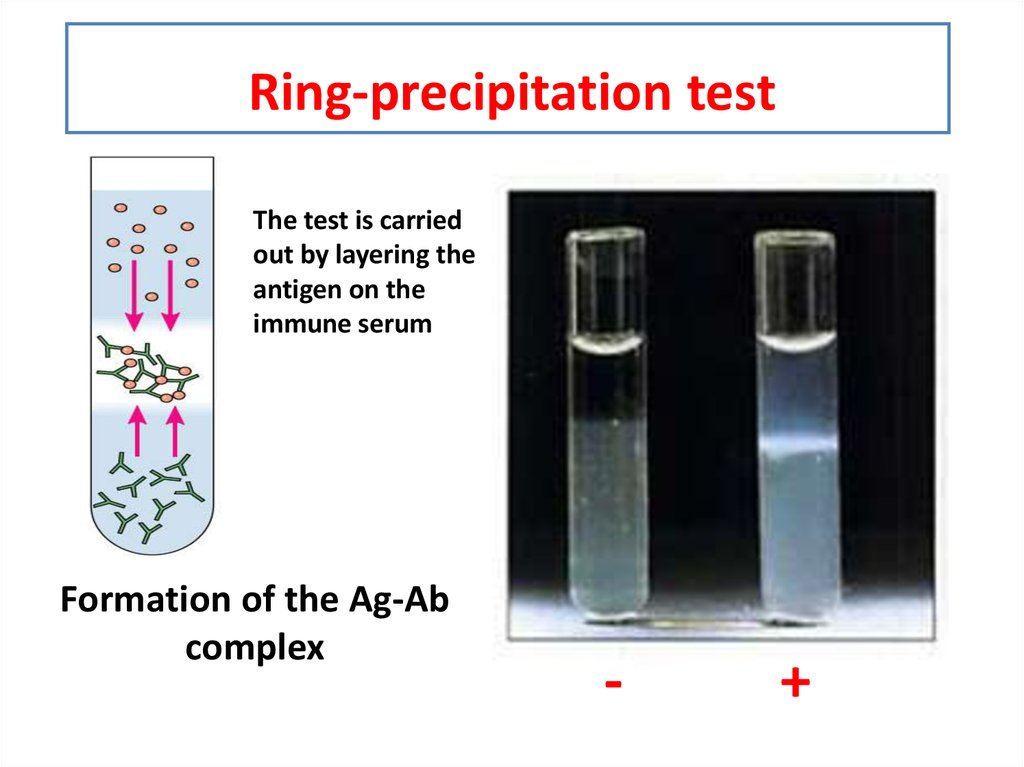
In the qualitative analysis procedure, the chemical properties of an unknown substance are determined by systematically reacting the unknown with a number of different reagents. Unlike covalent compounds, which can be identified using physical properties like boiling point and refractive index, ionic compounds are more appropriately identified with their chemical properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
